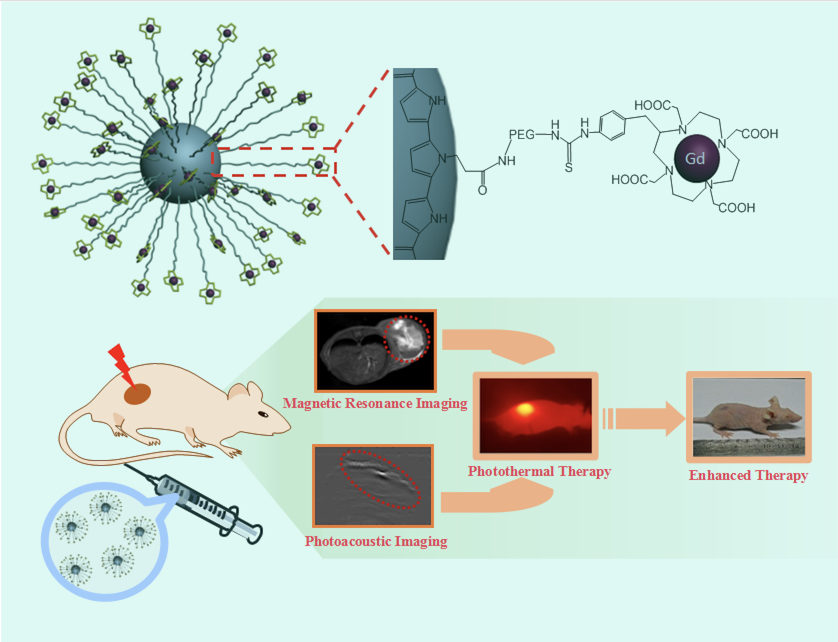
磁共振成像可提供解剖细节和高质量的软组织三维图像,但其灵敏度较低。将磁共振和光声这两种高度互补的成像融合可获得更多、更全面的信息,从而实现精准光热治疗。戴志飞课题组通过聚乙二醇将四氮十二环四乙酸钆连接到聚吡咯纳米粒子上,静脉注射后能选择地附集在肿瘤部位,导致光声和T1磁共振信号均显著增强,使肿瘤组织和正常组织明显地区分开来,从而引导近红外激光准确照射肿瘤部位,两周后实现了肿瘤的完全消融,并减少了对肿瘤周围正常细胞的损伤。相关结果发表在Adv. Funct. Mater. (2015, 25, 1451–1462),并被选为“亮点”推荐。梁晓龙副研究员为该文的第一作者。
以上工作得到国家杰出青年科学基金和国家自然科学重点基金的资助。
Polypyrrole nanoparticles conjugating gadolinium chelates were successfully fabricated for dual-modal magnetic resonance imaging (MRI) and photoa- coustic imaging guided photothermal therapy of cancer, from a mixture
of pyrrole and pyrrole-1-propanoic acid through a facile one-step aqueous dispersion polymerization, followed by covalent attachment of gadolinium chelate, using polyethylene glycol as a linker. The obtained PEGylated poly- pyrrole nanoparticles conjugating gadolinium chelates (Gd-PEG-PPy NPs), sized around around 70 nm, exhibited a high T1 relaxivity coefficient of 10.61 L mM-¹s-¹, more than twice as high as that of the relating free Gd3+ complex (4.2 L mM-¹s-¹). After 24 h intravenous injection of Gd-PEG-PPy NPs, the tumor sites exhibited obvious enhancement in both T1-weighted MRI intensity and photoacoustic signal compared with that before injection, indicating the efficient accumulation of Gd-PEG-PPy NPs due to the introduc- tion of the PEG layer onto the particle surface. In addition, tumor growth could be effectively inhibited after treatment with Gd-PEG-PPy NPs in combination with near-infrared laser irradiation. The passive targeting and high MRI/photo- acoustic contrast capability of Gd-PEG-PPy NPs are quite favorable for precise cancer diagnosing and locating the tumor site to guide the external laser irra- diation for photothermal ablation of tumors without damaging the surrounding healthy tissues. Therefore, Gd-PEG-PPy NPs may assist in better monitoring the therapeutic process, and contribute to developing more effective “personal- ized medicine,” showing great potential for cancer diagnosis and therapy.